Spiegelberg GmbH & Co. KG
Medical technology since 1986
Committed to progress
Spiegelberg, based in Hamburg, manufactures and sells innovative neurosurgical products and is a true pioneer in the field of "Technology for brains". We wish to apply our enthusiasm for innovations and our passion for medical technology, which is always aligned to the needs of our customers, to simplifying your daily medical work – whether it is with the measurement of intracranial and intraabdominal pressure or the treatment with external ventricular, subdural or lumbal drainage.
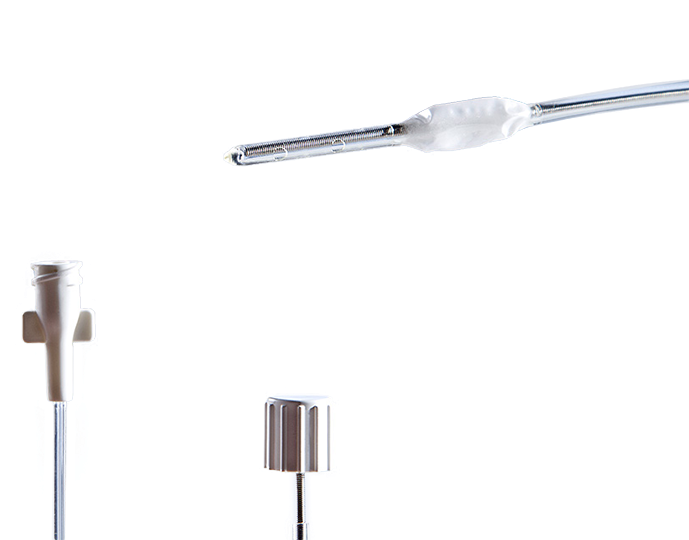
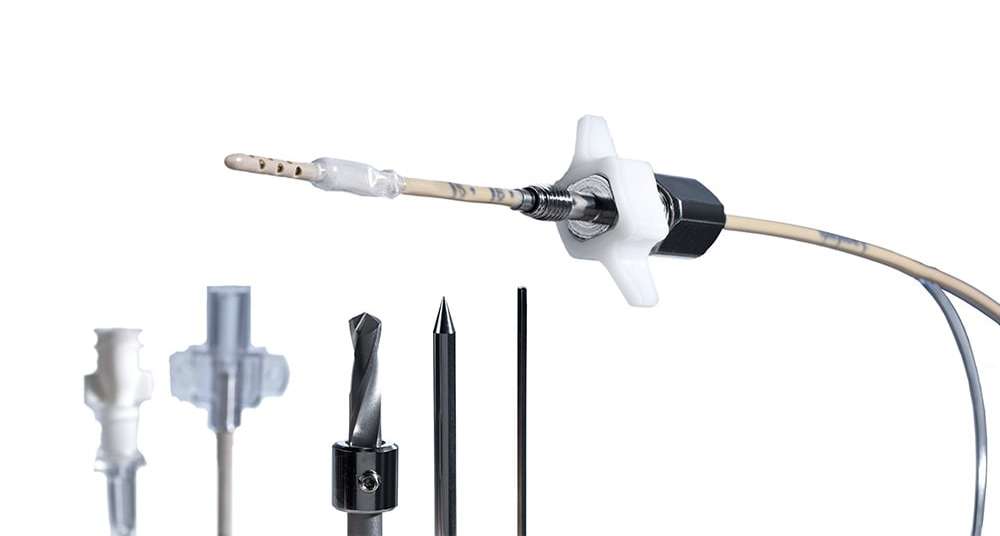
Innovative neurosurgical products: monitors, probes, catheters & EVD kits
Our close cooperation with well-known research institutions and leading hospitals and specialists in the field of neurosurgery enables us to supply our customers with high-quality medical devices such as ICP monitors, ICP probes, catheters & EVD kits, and IAP catheters along with accessories.
All our designs, manufacturing, services and sales of intracranial pressure measuring devices, neurosurgical medical devices and additives are based on the DIN EN ISO 13485 certificate.
World-wide accredited
Spiegelberg was established in 1986 in Hamburg and today supports customers in more than 60 countries where medical services rely on our innovative neurosurgical devices and the quality of our research and manufacturing – Made in Germany.
Spiegelberg is part of the Luciole Medical AG
In the summer of 2023, Luciole Medical AG, a medical technology company from Switzerland, acquired Spiegelberg GmbH. Both companies are now working together to expand their position as leading manufacturers in the field of brain monitoring.
Latest News
Good to know
This is why Spiegelberg is the choice:
Certified quality management system
Our quality management system certified by DIN EN ISO 13485 guarantees controlled development influenced by the requirements of customers and the manufacture of our products under closely monitored conditions. This guarantees that they are safe for users and patients.
Innovation prize awarded
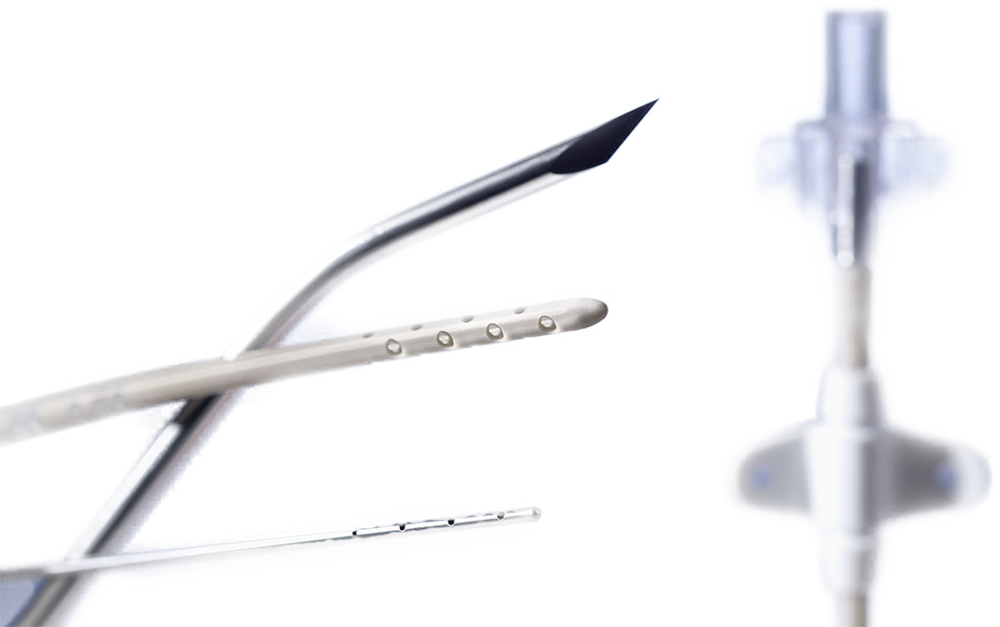
Spiegelberg was awarded first prize in the competition for the Innovation Prize awarded by the senate of the city of Hamburg (Innovationspreis des Senats der Freien und Hansestadt Hamburg) for the development of an “antimicrobial ventricular catheter”. The plastic coating of Silverline® catheters contain a patented silver additive that reduces the possibility of microbial colonisation of the surface of the catheter.